15 years one-stop China custom CNC machining parts factory
 139 |
Published by VMT at Feb 23 2026 | Reading Time:About 2 minutes
139 |
Published by VMT at Feb 23 2026 | Reading Time:About 2 minutes
Stress Corrosion Cracking (SCC) — frequently occurring in metals such as cartridge brass, 7075-T6 aluminum, and austenitic stainless steel (SS304) — is a phenomenon where materials or finished components can fail abruptly under high temperatures or chemical environments like chloride exposure. These cracks can be insidious; some remain internal and completely invisible to the naked eye. Understanding which materials are prone to SCC, which offer superior stability, and how to effectively detect and prevent these failures is critical for your engineering design and manufacturing success. Read on to dive deep into the essentials of Stress Corrosion Cracking.

Stress Corrosion Cracking (SCC) is the growth of crack formation in a corrosive environment. It is the result of three critical factors acting simultaneously:
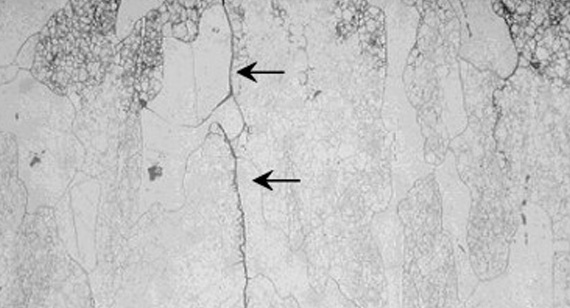
Unlike general corrosion which thins the metal, SCC produces fine, branching, or "spider-web-like" cracks. These cracks can be intergranular (following grain boundaries) or transgranular (cutting through grains), leading to sudden brittle failure.
If you plan to design or manufacture your metal components which will be used in environments like high-temperature, humidity, salt spray or with chemicals of ammonia, chlorides, etc., you'd better to avoid use materials that are prone to SCC to avoid your "parts failure in few months or suddenly " . Below table lists common materials that are prone to Stress Corrosion Cracking:
Table 1: Prone to Stress Corrosion Cracking Metals
| Material Category |
Susceptible Grades |
Triggering Environment |
Failure Mechanism |
| Copper Alloys (Brass) |
C26000 (Cartridge Brass), C28000 | Ammonia and its derivatives | Known as "Season Cracking"; occurs even with trace amounts of ammonia. |
| High-Strength Aluminum |
7075-T6, 2024-T3 | Chlorides, Humidity, Salt Spray | Common in aerospace; T6 tempers are particularly vulnerable to intergranular cracking. |
| Austenitic Stainless Steel |
SS304, SS316 | Chlorides + High Temp (>60°C) | The most common industrial SCC; leads to "spider-web" branching cracks. |
| High-Strength Carbon Steel |
AISI 4140, 4340 | Hydrogen Sulfide (H2S), Caustics | Known as Sulfide Stress Cracking (SSC); a major risk in "sour" oil and gas wells. |
| Magnesium Alloys |
AZ31B, AZ91 | Distilled Water, Chlorides | Highly reactive; even mild moisture can trigger cracking under high tensile stress. |
| Titanium Alloys |
Ti-6Al-4V (Grade 5) | Methanol, Fuming Nitric Acid | While generally resistant, halides and high-temperature salts can cause sudden failure. |
Complementary Explain Types of Stress Corrosion Cracking
While "Stress Corrosion Cracking" is the umbrella term, several specialized forms target specific industries:
Materials that have specific chemical compositions (like high nickel content) or unique microstructures (like dual-phase grains) can block crack propagation; and you can choose such materials to prevent SCC at the design stage:
Table 2: Materials that Offer Good Stability to Resist SCC
| Material Category |
Notable Grades |
Key Advantage |
Typical Applications |
| High Nickel Alloys |
Inconel 625, Hastelloy C-276 | Nickel content >45% makes them virtually immune to chloride SCC. | Aerospace engines, chemical reactors, deep-sea valves. |
| Duplex Stainless Steel |
2205, 2507 (Super Duplex) | A 50/50 mix of austenite and ferrite; the ferrite phase acts as a physical barrier to cracks. | Desalination plants, offshore oil platforms. |
| Ferritic Stainless Steel |
SS 444, SS 430 | Contains little to no nickel; the BCC crystal structure is inherently resistant to chlorides. | Solar water heaters, heat exchangers, automotive trim. |
| Copper-Nickel Alloys |
90/10 or 70/30 CuNi | Unlike brass, these contain no zinc, eliminating the risk of ammonia-induced "Season Cracking." | Marine condensers and piping systems. |
| Low-Strength Carbon Steel |
Standard Grade B Carbon Steel | When kept below 22 HRC (Rockwell C), these steels are much more stable in H2S environments. | General oilfield piping (compliant with NACE MR0175). |
Detecting Stress Corrosion Cracking (SCC) is difficult if only through naked eyes because the damage often begins at a microscopic level of the metal. Unlike general rust, which is easy to see, SCC can hide beneath a good clean-looking surface until the moment of catastrophic failure.
Visual inspection is the first basic check, but it is typically only effective during the later stages of cracking. You should look for "spider-web" or branching patterns on the surface, especially near welds or areas of high mechanical load. This method is suitable for routine daily checks of large structures, but it cannot be relied upon for critical safety components where cracks might be internal.
For a more reliable diagnosis, Liquid Penetrant Testing (PT) is used when you need to find surface-breaking cracks that are too small for the naked eye. This is ideal for non-porous metals like stainless steel or aluminum after they have been cleaned. It is best performed during scheduled maintenance shutdowns or after a component has been exposed to a known corrosive leak.
When the integrity of the entire part is at stake, Ultrasonic Testing (UT) or Eddy Current Testing is necessary. These methods use sound waves or electromagnetic fields to "see" through the metal. You should choose this high-level detection in daily maintaining check if you are working with thick-walled pressure vessels, aerospace landing gear, or high-pressure piping where internal cracks could lead to explosions or structural collapse.
Reducing the risk of SCC requires a proactive approach that starts during the manufacturing phase and continues through the part's operational life. Here is how you can mitigate these risks in different scenarios:
1. If Welding is Required
Welding is a major contributor to SCC because it introduces extreme local heat and leaves behind residual tensile stress. To reduce risk, always perform Post-Weld Heat Treatment (PWHT) or stress-relief annealing. This "relaxes" the metal grains and neutralizes the internal pull that causes cracks to grow. Additionally, using low-hydrogen electrodes and ensuring proper weld geometry can prevent the formation of "stress raisers" where cracks typically start.
2.If CNC Machining is Required

In precision machining, the goal is to avoid leaving the surface in a "stretched" (tensile) state. At VMT, we reduce SCC risks by using ultra-sharp cutting tools and optimized feed rates to ensure the metal is sheared cleanly rather than "torn." Aggressive, dry machining creates heat and surface tension; therefore, high-pressure flood cooling is essential to maintain material stability. For high-risk parts, we recommend Shot Peening after machining. This process hammers the surface to create a layer of compressive stress, which acts as a physical shield that prevents cracks from ever opening.
3. Avoid Incompatible Environments for Prone Materials
For Austenitic Stainless Steels (304/316): Avoid environments with high chloride concentrations (like salt spray or brackish water) if the temperature exceeds 60°C.
For Copper/Brass: Ensure the material is not exposed to ammonia, which is often found in cleaning agents or agricultural fertilizers. If the environment cannot be changed, you must switch to more stable materials like Duplex Stainless Steel or Inconel.
4. Implement Surface Barriers and Inhibitors
If you must use a susceptible material in a harsh environment, use protective coatings or electrochemical barriers. Applying anodizing to aluminum or specialized epoxy coatings to steel can prevent corrosive ions from reaching the metal surface. In closed-loop systems, such as boilers or cooling towers, adding chemical corrosion inhibitors to the water can neutralize the environment's aggressiveness, effectively "starving" the SCC process before it begins.
5. If High-Strength Heat Treatment is Required (e.g., T6 vs. T651)
In materials like 7075 or 6061 aluminum, the T6 temper (solution heat treated and artificially aged) provides maximum strength but often leaves behind high levels of internal residual stress from the rapid quenching process. However, in the T651 temper, the material is stress-relieved by controlled stretching after quenching but before aging. While this might result in a very slight sacrifice in absolute hardness or strength, it significantly stabilizes the metal’s internal structure, drastically reducing the risk of sudden cracking during or after machining.
6. If Cold Working is Involved (Bending, Rolling, or Forging)
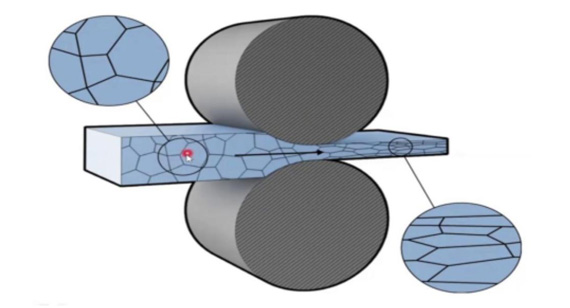
Cold working processes—such as bending, rolling, or cold forging—increase the dislocation density within the metal’s grain structure, creating areas of high residual tensile stress. To reduce SCC risk during these operations, you should:
Stress Corrosion Cracking (SCC) is an important factor that you must take into consideration; From initial material selection and part design to production and long-term maintenance, this should be carefully managed. In this guide, we have broken down the fundamental basics of Stress Corrosion Cracking and identified which materials are high-risk versus those that offer good stability. We also outlined the essential methods for detecting microscopic cracks before they lead to catastrophic failure, along with actionable prevention strategies in different situations. Understanding these is not just about avoiding material failure, but also ensuring the reliability, safety, and longevity of your parts or components.
A client in the offshore energy sector used SS316L for a small, precision-machined hydraulic sensor block. These blocks operated in a high-temperature (80°C), high-chloride marine environment. Despite the material’s general corrosion resistance, the blocks experienced "spider-web" micro-cracking around the internal threaded ports within 6 months, leading to high-pressure leaks. The failure was traced to Chloride Stress Corrosion Cracking (CLSCC), exacerbated by high residual stresses left over from aggressive tapping and cold-work-inducing machining passes.
VMT CNC Machining Factory Solution
The Result
The redesigned Duplex 2205 sensor blocks have been deployed in subsea conditions for over 30 months without a single report of cracking or pressure loss. By combining a material upgrade with stress-aware CNC machining, VMT extended the component's service life by over 500%.

Q1: Is 316 stainless steel immune to SCC?
No. While 316 is more resistant than 304, it is still susceptible to chloride stress corrosion cracking at elevated temperatures (typically above 60°C).
Q2: What is the most effective way to stop SCC in existing designs?
If the material cannot be changed, introducing compressive stress via shot peening or applying cathodic protection (sacrificial anodes) are the most effective mitigation strategies.
Q3: Does surface roughness affect SCC?
Yes. A rougher surface provides more "pits" and "notches" for corrosive ions to accumulate, accelerating crack initiation. High-precision CNC finishing is essential.
Q4: Can SCC occur in carbon steel?
Yes, particularly in the form of caustic stress corrosion cracking or sulfide cracking in oil and gas environments.
Q5: What is the difference between SCC and Hydrogen Embrittlement?
While both lead to brittle failure, SCC requires a corrosive environment, whereas Hydrogen Embrittlement is caused by the presence of hydrogen within the metal lattice, often introduced during plating or welding.
Q6: What ASTM standards govern SCC testing?
Common standards include ASTM G30 (U-bend test) and ASTM G36 (Boiling Magnesium Chloride test) for stainless steels.
